What is air pollution ?
A condition of the air, arising wholly or partly from the presence therein of one or more air contaminants, that endangers the health, safety, or welfare of persons, interferes with normal enjoyment of life or property, endangers the health of animal life, or causes damage to plant life or property. Pollutants can be solid, liquid or gas.

Paris under smog – Source: Flickr
Sources of pollution
Sources of air pollution are varied and may be anthropogenic (man-made) or natural. The main anthropogenic sources are:
- burning of fossil fuels in electricity generation, transport, industry and households;
- industrial processes and solvent use, for example in chemical and mineral industries;
- agriculture;
- waste treatment.
Natural causes of air pollution include volcanism, dust storms, forest fires and gases such as methane, emitted by the digestion of food by animals, for example cattle. Today even the natural causes of air pollution are largely determined by man: the production of biogenic methane significantly depends on the size of the herd (which is controlled by humans), forest fires are often lit by man and the withering of the Sahel in Africa, a result of global warming, can also lead to more dust storms.
On top of that Europe may also experience pollution coming from other countries, since air pollutants can be transported over long distances.
Which pollutants are we talking about ?
 In Europe, emissions of many air pollutants have fallen substantially since 1990. However, since 1997, measured concentrations of particulate matter and ozone in the air have not shown any significant improvement. A significant proportion of Europe’s urban population still lives in cities where EU air quality limits (set for the protection of human health) are exceeded. The need to reduce exposure to air pollution remains an important issue.
In Europe, emissions of many air pollutants have fallen substantially since 1990. However, since 1997, measured concentrations of particulate matter and ozone in the air have not shown any significant improvement. A significant proportion of Europe’s urban population still lives in cities where EU air quality limits (set for the protection of human health) are exceeded. The need to reduce exposure to air pollution remains an important issue.
There are three pollutants that make up for the major part of the pollution in Europe: fine particulate matter (PM), ground-level ozone (O3) and Nitrogen Dioxide (NO2).
Particulate matter is the sum of all solid and liquid particles suspended in air, many of which are hazardous. This complex mixture includes both organic and inorganic particles, such as dust, pollen, soot, smoke, and liquid droplets. These particles vary greatly in size, composition, and origin.
Ozone (O3) is a gas that is present in two layers of the atmosphere. In the stratosphere (between about 12 and 50 km altitude above the surface) ozone forms a layer that shields the Earth from ultraviolet rays. At ground level (in the troposphere) however, ozone is considered as a major air pollutant. Ground-level ozone is formed from other pollutants and can react with other substances, in both cases under the action of light.
Nitrogen oxides (NOx) are produced as a result of road traffic and other fossil fuel combustion processes. They are also precursors for a number of harmful secondary air pollutants, such as ozone, nitric acid, and nitrates, which contribute to increased respirable particle levels in the atmosphere.
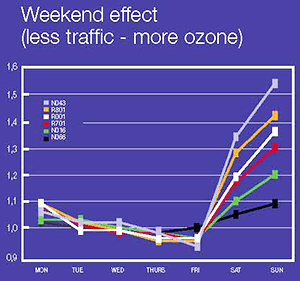
Ozone concentrations are often low in busy urban centres and higher in suburban and adjacent rural areas, particularly on sunny days in summer. And concentrations are higher during the weekend than during weekdays. The reason for this is the fact that nitric monoxide, which is produced in large quantities by intense traffic, actually breaks down ozone by reacting with it. In cities, where traffic is fare more intense than in the countryside, ozone is broken down more frequently. And another factor that should not be forgotten is the fact that ozone and its precursors can be transported through air over hundreds of kms.
Effects
Two of these three pollutants – PM and O3 – are now generally recognised as the most significant in terms of health impacts.
Long-term exposure to current ambient particulate matter concentrations may affect the lungs of both children and adults and may reduce life expectancy by a few months, mainly in subjects with pre-existing heart and lung diseases.
Ozone is also known to cause adverse health effects. Short-term exposure to ozone peaks can temporarily affect the lungs, the respiratory tract, and the eyes. It can also increase the susceptibility to inhaled allergens. Long-term exposure to relatively low concentrations of ozone can reduce lung function.
Long-term and peak exposure to PM and O3 thus can lead to a variety of health effects, ranging from minor effects on the respiratory system to premature mortality. Since 1997, up to 45% of Europe’s urban population may have been exposed to ambient concentrations of particulate matter above the EU limit set to protect human health; and up to 60% may have been exposed to levels of ozone that exceed the EU target value. It has been estimated that PM2.5 (fine particulate matter with a diameter smaller than 2.5 µm) in air has reduced statistical life expectancy in the EU by more than 8 months.
As for PM and O3, the evidence of nitrogen dioxide (NO2) on health comes from different sources of information, including observational epidemiology, controlled human exposures to pollutants and animal toxicology. The evidence for acute effects of NO2 comes from controlled human exposure studies to NO2 alone. For the effects observed in epidemiological studies, it is difficult to establish those attributable to NO2 alone, because the presence of NO2 is closely linked to the formation or presence of other air pollutants.>
NOx plays also a role in the formation of acid rain.
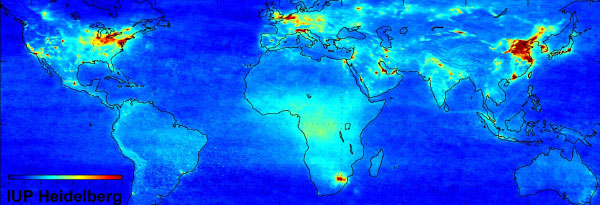
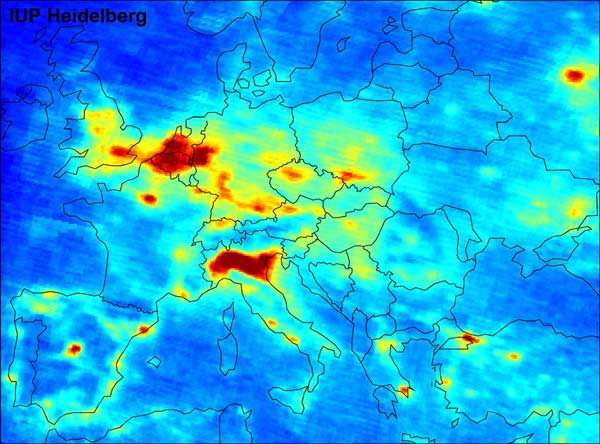
Global air pollution map produced by Envisat's SCIAMACHY and detail from this map showing Europe pollution.
The map illustrates regions where traffic and fuel combustion contribute to NO2 air pollution.
It shows the mean ground level nitrogen dioxide (NO2) concentration between January 2003 and June 2004,
as measured by the SCIAMACHY instrument on ESA's Envisat. Credits: University of Heidelberg
Air pollution also damages our environment, for example by causing:
- Acidification: acid depositions (e.g. acid rain) have harmful effects on plants, aquatic animals, and infrastructure. Acid rain is mostly caused by emissions of sulphur and nitrogen compounds which react in the atmosphere to produce acids. The gases can be carried hundreds of kilometres in the atmosphere before they are converted to acids and deposited. Since 1980 the sensitive ecosystems that are subject to excess acid deposition have substantially fallen. Nevertheless in 2010 more than 10% of all European natural ecosystems will still be subject to acid deposition beyond their critical load (the threshold above which the ecosystem can no longer bear environmental load without significant damage);
- Eutrophication: is an increase in the concentration of chemical nutrients in an ecosystem to an extent that increases in the primary productivity of the ecosystem. In fresh water environments, eutrophication is almost always caused by phosphates, since phosphorus is the substance that usually limits biological growth in fresh water. On land and in the sea, however, it is nitrogen that is the limiting factor in the majority of cases. The deposition of nitrogen - originating from emissions of nitrogen oxides and ammonia - therefore acts as a fertilizer in nature. Less progress has been made in reducing eutrophication. More than 40% of sensitive ecosystem areas are subject to nitrogen deposition beyond their critical loads;
- Crop damage: ground-level ozone forms a threat to agriculture since it impairs photosynthetic processes, resulting in significant reduction in agricultural yields. A significant proportion of the crops, particularly in southern, central and Eastern Europe, is exposed to ozone levels above the EU target value.
Measures
In addition to the development of new technologies, there are various air pollution control techniques and land use planning strategies available to reduce air pollution. Planning could limit itself to zoning and transport infrastructure planning, but it can play an important role in the distribution of land use types by looking for the optimal locations for both economy and ecology.
transport infrastructure planning, but it can play an important role in the distribution of land use types by looking for the optimal locations for both economy and ecology.
Pollution from mobile sources might be reduced by diminishing the allowed emission values for classical vehicles (cars, aeroplanes ...), by expanding these regulations to new pollution sources (such as cruise ships, lawn trimmers, snowmobiles ...), by increasing fuel efficiency, by conversion to cleaner fuels and/or by conversion to electric vehicles.
Apart from that, legal regulations can also be applied. A report from the European Environment Agency shows that road transport remains Europe’s single largest air polluter. Several directives are installed to regulate this problem.
A third factor, and certainly one not to be forgotten, is the role of the public in the battle against air pollution. While legal regulations certainly have an important top-down impact on the use of pollutants, people should be made more aware of their own (bottom-up) influence on air quality. Their impact does not lie in giant measures taken by a few actors, but in the power of the masses: one less car trip per person per year can already have a tremendous effect on the environment. Imagine what else 800 million people can do without much of an effort...
Sources
Air Pollution - The Environment: A Global Challenge
Air pollution and daily mortality in the Netherlands over the period 1992 - 2002 - RIVM - The Netherlands
Air Pollution and Respiratory Health - CDC
Wikipedia Air pollution
This page was written in 2009, as additional information to the poster series "10 years of Imaging the Earth"